Core Values

Consulting for Growth Business Strategy
After more than 14 years in the medical device industry, we saw the same struggle over and over again — great companies with incredible technologies facing endless hurdles just to bring their products to new markets. Many had the passion, innovation, and quality to make a real difference in patients’ lives, but they were held back by one thing: the complexity of regulatory pathways and the lack of trustworthy local support.
That’s when Teralis was born — out of a simple yet powerful belief: every good product deserves a clear path to the people who need it most.
We built Teralis to bridge that gap — guiding medical device/ IVD manufacturers and consumer product companies through every stage of registration, approval, and market access across the Middle East and Africa. Our approach is personal and precise. We don’t just help you meet regulations; we make sure you understand them, comply confidently, and never face unnecessary roadblocks again.
And because we’ve seen how critical local partnerships can be, we go beyond consulting — helping you connect with trustworthy distributors who represent your brand with integrity and align with your long-term goals.
At Teralis, we’re not just consultants. We’re your regional partners — committed to making your expansion seamless, compliant, and successful. With us, you’ll never have to second-guess your next step.
Founder & CEO
Syam Sasidharan
Dedicated Team Members
100% Clients Saticfaction
Delivering Results
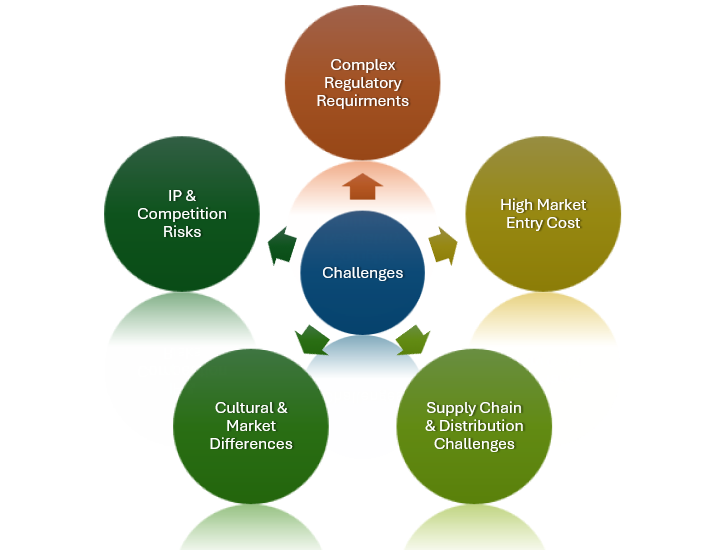
Transforming Challenges Into Scalable Opportunities
Medical Device & IVD Registration
Software as Medical Device Registration
Regulated Consumer Products Registration
Type Approval
Advantages Of Expanding Your Business
-
Access to GCC Market
-
Wider Customer Base
-
Unlimited Opportunities







