We Help People & Businesses
Teralis
Medical Device/ IVD Registration
We specialize in guiding medical device and IVD companies through the complex regulatory landscape of the Middle East. From product classification, dossier preparation, and registration to labeling compliance, approvals, and post-market surveillance — we streamline your path to market. With deep expertise in local regulations, authorities, and compliance expectations, we help you minimize delays, reduce risks, and ensure your innovations — from medical devices to diagnostic kits — reach healthcare providers and patients faster across the region.
SaMD Registration
The rise of digital health has made Software as a Medical Device (SaMD) one of the fastest-growing segments in the healthcare sector — and regulators across the Middle East are evolving to meet this innovation. At Teralis, we help you navigate the complex SaMD registration pathways in the GCC and broader Middle East region, ensuring your software meets both medical device and cybersecurity standards.
We support you through every step — from product classification and risk assessment to technical documentation, and conformity assessment. Whether your software supports diagnosis, treatment, or remote monitoring, we ensure it aligns with global frameworks like IMDRF, MDR 2017/745, and local GCC guidance, positioning you for faster registration and regional market access.
Examples of SaMD solutions we support:
AI-based diagnostic and imaging analysis software
Clinical decision support systems (CDSS)
Remote patient monitoring and telehealth platforms
Digital therapeutics and wellness-to-medical-grade app transitions
We bridge technology and compliance — enabling your digital innovation to safely and confidently enter the Middle East healthcare market.


Teralis
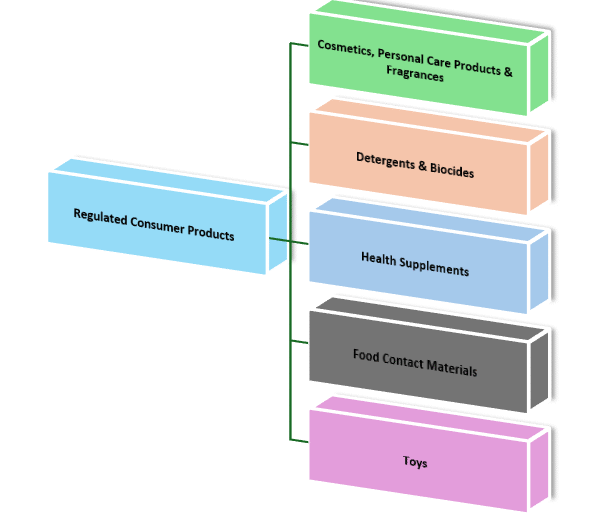
Regulated Consumer Products Registration
We help you register your cosmetics, personal care products, and fragrances with Dubai Municipality, ensuring full compliance with local regulations. Our service also assists businesses in obtaining the necessary trade licenses to legally sell and import these products across the UAE. We make sure your consumer products meet all UAE safety and labeling regulations, so you can confidently sell your products across the Emirates.
Type Approval
Entering the Middle East market requires more than just great technology — it demands full compliance with each country’s telecom and regulatory standards. At Teralis, we help companies obtain Type Approval for all kinds of devices that use wireless, radio frequency, or telecommunication technology — whether medical or non-medical.
We handle the entire process — from documentation and testing coordination to communication with local authorities — ensuring your products meet regional standards and are ready for sale or distribution.
We support a wide range of devices, including:
Medical Devices: Remote patient monitoring systems, wireless diagnostic equipment, wearable medical sensors, and hospital communication devices.
Non-Medical Devices: Wireless routers, smart home systems, IoT devices, GPS trackers, RFID-based equipment, and other connected electronics.
Examples:
Medical devices that use radio frequencies to transmit data, such as some remote patient monitoring devices or certain types of diagnostic equipment.
Devices such as implantable pumps, pacemakers/defibrillators, adjustable implants or sensors that use RF telemetry, Bluetooth or other wireless to communicate with external programmer/control units or patient devices.
A wireless insulin pump that receives glucose data via RF from a sensor and then communicates with clinician software or smartphone. Because they emit/receive radio waves, they must comply with radio equipment directives and spectrum/EMC rules (type approval) and meet the medical device regulatory pathway for safety/clinical performance.
Medical IOT modules, RFID tagged diagnostic kits or instrument modules
Devices that are implanted and use wireless charging (inductive or RF) and may also transmit telemetry data. For example: ultra-low-power active medical implants operating at specific frequency bands in the 401-406 MHz range





